LAL Endotoxin Test Kit Kinetic Turbidimetric Method
Min.Order : 1 Pieces Quick Quotation >
Conpany Profile
Product Details
LAL endotoxin test kit kinetic turbidimetric method

LAL endotoxin test kit kinetic turbidimetric method
We supply kinetic turbidimetric LAL lysate for pharmaceutical and medical device industries for testing the endotoxin level of the injectable drugs and implantable medical devices at China. In the past 35 years, we devoted ourselves to the research of LAL endotoxin testing technology, to make sure the quality of drug products and medical devices, allows the monitoring of the endotoxin level during production process.
1. Product introduction
Kinetic turbidimetric endotoxin test kit is the endotoxin assay kits by kinetic turbidimetric method. Based on the principle that in the presence of endotoxin (pyrogen, lps) LAL becomes turbid, the increase rate of turbidity is proportional to the endotoxin concentration of the sample, the endotoxin is quantified. The kit includes kinetic turbidimetric LAL reagent, Control Standard Endotoxin, LAL Reagent Water (Water for Bacterial Endotoxin Test), Constitution Buffer, pyrogen free pipette tips, pyrogen free glass test tube for control standard endotoxin dilution and pyrogen free microplate. The detection limit for the assay kit is 0.005EU/ml to 50EU/ml. Kinetic turbidimetric endotoxin assay require a kinetic microplate reader such as ELx808IULALXH or a kinetic tube reader. And kinetic software is also required for calculate the endotoxin concentration.
2. Product parameter
Assay range: 0.005-50EU/ml
1.7ml per vial or 2.2ml per vial
3. Product application
End-product endotoxin (pyrogen) qualification, Water for injection endotoxin assay, raw material endotoxin testing or endotoxin level monitoring during manufacturing process for pharmaceutical companies or medical devices manufacturers.
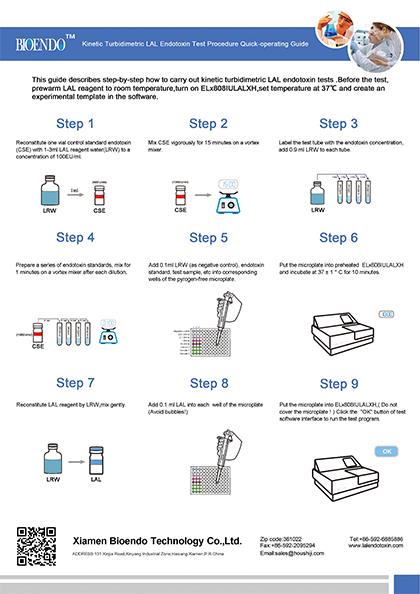
Operation chart
Standard curve:
4. Ordering information
Catalog Number | Description | Kit Contents |
KT9028 | Bioendo™ KT Endotoxin Test Kit (Kinetic Turbidimetric Assay), 2340 tests/kit | 90 Kinetic Turbidimetric TAL Reagent, 2.8ml (26 Tests/vial); 20 CSE; |
KT0828 | Bioendo™ KT Endotoxin Test Kit (Kinetic Turbidimetric Assay), 208 tests/kit | 8 Kinetic Turbidimetric TAL Reagent, 2.8ml (26 Tests/vial); 4 CSE; 2 TAL Reagent Water, 50ml/vial; |
KT0852 | Bioendo™ KT Endotoxin Test Kit (Kinetic Turbidimetric Assay), 400 tests/kit | 8 Kinetic Turbidimetric TAL Reagent, 5.2ml (50 Tests/vial); 4 CSE; 2 TAL Reagent Water, 50ml/vial; |
5. Product Qualification

6. Ordering FAQ
Product condition:
The LAL reagent sensitivity and the Control Standard Endotoxin potency are assayed against USP Reference Standard Endotoxin. The LAL reagent kits come with product instruction, Certificate of Analysis, MSDS.
How to order:
Please contact [email protected] to order the LAL reagents endotoxin detection kit. There is no minimal order volume required. The price depends on the order volume. We provide distributor discount for our distributors. The price is in US dollars. The final cost will be the order price plus the shipping cost. We need to know the shipping item to calculate the shipping weight.
Payment Method:
We required 100% prepayment for all orders. We accept T/T bank transfer or Paypal. If pay by Paypal 5% additional charge is required.
Shipping Condition:
Once we received the payment, we will ship in two days. Shipping is usually by air, it takes less than one week to reach the location. LAL Reagent is stable at controlled room temperature for more than two weeks. We usually ship the reagent at room temperature. After receipting LAL Reagent, long term storage at 2-8? is required.
Storage Condition:
The LAL reagent is stable for more than two years at 2-8?. Store LAL reagent at 2-8? , avoid light. LAL Reagent Water is stable for more than three years at 2-20? .
Note:
LAL Reagent ( TAL Reagent ) from our company are formulated from the aqueous extract of circulating amebocytes of Chinese horseshoe crab (Tachypleus tridentatus).
Recommend Product